Eagle Syndrome is best known for throat pain. Stylogenic Jugular Vein Compression is increasingly recognized as a cause of headaches, brain fog, and intracranial hypertension. But there is a third consequence of the same anatomical problem that almost no one talks about: palsy of the spinal accessory nerve, which causes progressive weakness of the trapezius — the large muscle that holds your shoulder up — and, in advanced cases, secondary Thoracic Outlet Syndrome (TOS).
The spinal accessory nerve is also called the 11th cranial nerve, or CN XI. It controls the trapezius and the sternocleidomastoid (the strap-like muscle on the front of the neck). It descends from the base of the skull and wraps around the internal jugular vein as both pass through the same narrow corridor. When that corridor closes, the nerve gets squeezed alongside the vein.
The trapezius slowly weakens. The shoulder blade starts to wing — to pop outward instead of staying flat against the ribcage. The shoulder gradually drops. And the bundle of nerves running from the neck into the arm — the brachial plexus — ends up stretched downward over the first rib. That sustained stretch is what produces the tingling, numbness, and arm pain that doctors call neurogenic Thoracic Outlet Syndrome.
A single anatomical abnormality at the base of the skull — most often the styloid process (a slender, finger-like bone that hangs from the skull behind the jaw) tilted inward, the bony wing of the topmost vertebra (the C1 transverse process) thickened, or both — can therefore produce symptoms in your hand and forearm, far from where the actual problem lies.
The Third Face of Eagle Syndrome
Most descriptions of Eagle Syndrome mention two clinical pictures:
- Classic Eagle Syndrome — throat pain, ear pain, and swallowing difficulty caused by irritation of the glossopharyngeal nerve and other nerves in the throat.
- Vascular Eagle Syndrome / Stylogenic Jugular Vein Compression — head pressure, headaches, pulsatile tinnitus, and brain fog caused by compression of the internal jugular vein.
The third face is CN XI palsy with secondary Thoracic Outlet Syndrome. It was formally described in 2023 by Mitchell et al., who called it “Winged” Eagle’s Syndrome — named for the way the shoulder blade wings outward as the trapezius wastes. 1 Two patients in their report had progressive shoulder weakness, lateral scapular winging, and trapezius atrophy, with imaging confirming spinal accessory nerve compression in the atlanto-styloid space at the base of the skull. Both improved after styloidectomy with nerve decompression — surgical removal of the styloid process combined with freeing the trapped nerve.
Eagle Syndrome Neurological Symptoms: An Overview
Eagle Syndrome can compress or irritate several different nerves at the base of the skull, and each produces its own pattern of symptoms:
- The glossopharyngeal nerve (CN IX) — throat pain, ear pain, swallowing difficulty.
- The vagus nerve (CN X) — vague chest discomfort, palpitations, abdominal symptoms.
- The sympathetic chain — facial pain, eye pain, referred pain.
- The spinal accessory nerve (CN XI) — the subject of this article. Trapezius weakness, shoulder droop, scapular winging, and downstream Thoracic Outlet Syndrome.
For the broader picture of how Eagle Syndrome affects nerves and symptoms across all of these patterns, see Eagle Syndrome: A Detailed Review. This article focuses on the cascade driven by CN XI compression specifically, because it is the most under-recognized — and the most likely to send a patient through the wrong workup.
Why the Spinal Accessory Nerve Gets Caught at the Skull Base
The atlanto-styloid space is the narrow corridor between the styloid process and the transverse process of the first cervical vertebra (C1, the atlas). When the styloid is angled inward against C1, that corridor narrows into a bony nutcracker.
The internal jugular vein — the main vein that drains blood from your brain — runs through that corridor. So does the spinal accessory nerve, which wraps around the vein as both descend from the jugular foramen at the base of the skull.
A detailed cadaveric study by Roberts and Cardozo (2024) showed something striking: in 56–96% of people, CN XI crosses in front of the internal jugular vein as both descend from the skull. In about 2.8% of people, the nerve actually passes through a hole in the wall of the vein itself (a fenestration). In rare cases (around 0.4%), the vein splits into two branches and the nerve runs between them. 2
The clinical implication is direct: in every one of these arrangements, the nerve and the vein are anatomically inseparable at the level of C1. You cannot squeeze one without squeezing the other.
The other lower cranial nerves passing through this region — the glossopharyngeal (CN IX), vagus (CN X), and hypoglossal (CN XII) — don’t share this intimate relationship with the vein and are typically spared. The internal carotid artery sits medial to the styloid, in a different compartment, and is also unaffected by this specific mechanism.
The patients most vulnerable are the ones with the rarer anatomy — particularly those whose nerve passes through a fenestration in the vein. For them, the nerve is essentially trapped inside the vein wall. Even modest narrowing at the skull base can be enough to compress it.
Why a ‘Normal’ Styloid Can Still Compress the Nerve
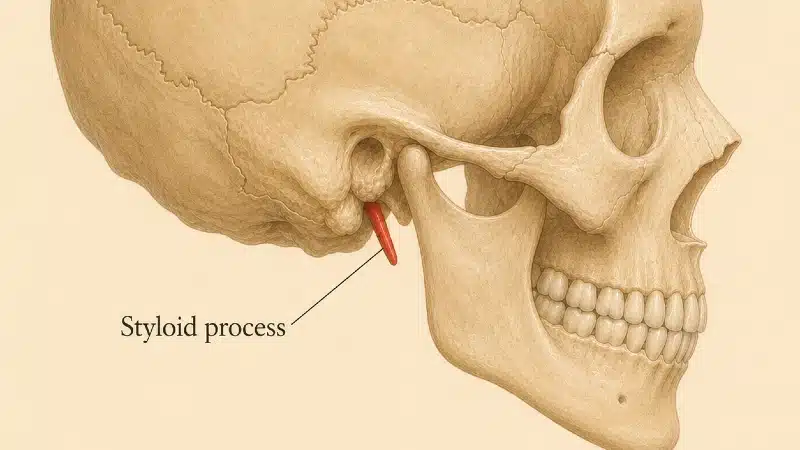
This is the same critical reframe as in the Jugular Vein Compression article: the styloid does not need to be long for any of this to happen.
In one of the original Mitchell case reports, the styloid measured only 2.2 cm — within normal limits — but it was angled 31 degrees in the coronal plane (tilted inward toward C1), and the atlanto-styloid corridor was narrowed down to just 1.5 mm. 1
What matters is the relationship between the styloid and the C1 transverse process, not styloid length. Stylo-atlantal syndrome is a more accurate term for this group of patients than classical Eagle Syndrome. The mechanism is mechanical compression in a bony nutcracker — the same mechanism that causes stylogenic jugular vein compression. The two conditions often coexist in the same patient.
If a CT report tells you “the styloid is normal length” and stops there, the report is incomplete. The atlanto-styloid distance must be measured.
Spinal Accessory Nerve Palsy: What CN XI Does, and Why You May Not Notice It Failing
The 11th cranial nerve is unusual: it is purely motor. It carries no pain fibers, no sensation, no autonomic input. It does only two things:
- It controls the trapezius — the large kite-shaped muscle that runs from the back of the skull, across the top of the shoulder, and down into your upper back. The trapezius holds the shoulder blade up against gravity, lifts the shoulder when you shrug, and stabilizes the scapula when you raise your arm.
- It controls the sternocleidomastoid (SCM) — the strap-like muscle on the front of the neck that turns your head toward the opposite side.
Because the nerve carries no pain fibers, compression of CN XI doesn’t hurt. The muscle just slowly weakens as it loses its nerve supply — a process doctors call denervation. The shoulder slowly drops. Many patients first notice it in old photographs — one shoulder lower than the other in family photos that span several years, getting more pronounced over time.
This is what makes spinal accessory nerve palsy so insidious. Eagle Syndrome’s classic throat pain announces itself loudly. Jugular vein compression announces itself with head pressure. CN XI compression creeps in.
The Cascade: From Skull Base to Fingers
The short version, in one sentence: a bone problem at the base of the skull slowly weakens the muscle that holds the shoulder up; the shoulder sags; and the nerves running to the hand get pulled taut over your first rib.
Here is the same cascade in detail, step by step, when the spinal accessory nerve is compressed at the atlanto-styloid space:
- Stylo-atlantal compression. The styloid process presses against C1, narrowing the atlanto-styloid corridor.
- CN XI is squeezed between the bony nutcracker and the internal jugular vein.
- The trapezius and SCM lose their nerve supply (denervation). The muscles begin to weaken and shrink.
- The shoulder loses its support — what doctors call loss of shoulder girdle suspension. The trapezius can no longer hold the collarbone and shoulder blade up against gravity.
- Shoulder droop develops. The hollow above the collarbone (the supraclavicular fossa) deepens visibly. The shoulder blade tilts outward — what doctors call lateral scapular winging.
- The space between the collarbone and first rib — the costoclavicular space — narrows as the collarbone descends.
- The brachial plexus gets stretched — what doctors call brachial plexus traction. The lowermost branches of those nerves — known as the lower trunk (C8–T1), which serve the pinky, ring finger, and the inner side of your forearm — get pulled downward across the first rib like a rope going taut over an edge.
- Secondary Thoracic Outlet Syndrome. Pain, tingling, and numbness (medically called paresthesias) along the inner side of the arm and the pinky-and-ring-finger side of the hand — what doctors call the medial arm and ulnar fingers — worse when you raise your arm overhead.
Each of these eight steps is documented in the medical literature. What is rare — and easy to miss — is that all eight can be present in a single patient, driven by a single anatomical problem at the skull base.
Trapezius Weakness, Scapular Winging, and Shoulder Droop: The Visible Signs
These are the signs that should make a doctor look upstream toward the skull base, rather than treating the arm symptoms in isolation.
Trapezius Weakness and Atrophy
The trapezius wastes slowly and asymmetrically. On the affected side, the muscle bulk above your collarbone looks visibly smaller or flatter than on the other side. The hollow above the collarbone (supraclavicular fossa) is deeper. When you shrug both shoulders against resistance, the affected side gives way first.
Many patients describe noticing it in the mirror over the course of months or years. Others are told by a physiotherapist or massage therapist that “one trap feels different.” A few are diagnosed only after a friend or family member points out an old photograph.
Scapular Winging
Scapular winging describes a shoulder blade that lifts off the back of the ribcage instead of sliding flat along it. There are several types of winging, and the specific pattern matters for diagnosis.
The pattern caused by CN XI palsy is called lateral scapular winging: the shoulder blade tilts outward and downward, most visible when you raise your arm forward or to the side. (This is different from the more familiar medial winging caused by long thoracic nerve injury, where the shoulder blade pops out toward the spine when you push against a wall.)
If your doctor or physiotherapist has noted scapular winging without identifying the cause, the question of which type matters. Lateral winging on the same side as a drooping shoulder, with no history of shoulder trauma or surgery, raises the question of CN XI involvement.
Shoulder Droop and Droopy Shoulder Syndrome
When the trapezius can no longer hold the shoulder girdle up, the shoulder gradually descends. One shoulder sits visibly lower than the other. In severe cases, the collarbone slopes downward toward the first rib at an unusual angle.
In 1984, Swift and Nichols described this pattern as Droopy Shoulder Syndrome — a form of Thoracic Outlet Syndrome caused by chronic shoulder droop alone, with no cervical rib, no scalene anomaly, no other obvious mechanical cause. 3 More recent work has reframed Droopy Shoulder Syndrome as a gateway condition: a precursor stage where the brachial plexus traction is still reversible, and where conservative treatment can interrupt the slide into permanent neurogenic TOS. 4
When the cause of that droop is spinal accessory nerve palsy from atlanto-styloid compression, the result is a cascading compression syndrome — a single chain of compression spanning skull base to thoracic outlet.
From Shoulder Droop to TOS: The Direct Evidence
The link between accessory nerve injury and Thoracic Outlet Syndrome is decades old. Most of the proof comes from a different scenario — what happens when CN XI is accidentally cut during routine neck surgery — but the cascade that follows is the same one this article describes. If a one-time injury to this single nerve can drive a patient all the way to TOS, chronic compression of the same nerve at the skull base can do the same thing.
In 2003, Al-Shekhlee and Katirji described two patients who developed full TOS after iatrogenic injury to CN XI — accidental damage during a routine cervical lymph node biopsy. 5 One ended up with complete occlusion of the axillary artery (the main artery into the arm) at 90 degrees of abduction — a vascular form of TOS. The other had electrophysiological evidence — measured by nerve conduction studies — of injury to the lower trunk of the brachial plexus, the same neurogenic pattern this article is built around. Both improved when the nerve was repaired and the shoulder was strengthened.
That single paper therefore documented both the vascular and the neurogenic faces of the downstream cascade, driven by the same upstream cause.
What’s new in the literature on Eagle Syndrome and TOS is the recognition that the same cascade can be driven from the opposite end — by chronic compression at the skull base rather than by acute surgical injury in the neck. The endpoint is the same. The starting point is different, and easier to miss.
When to Look Upstream Toward the Skull Base
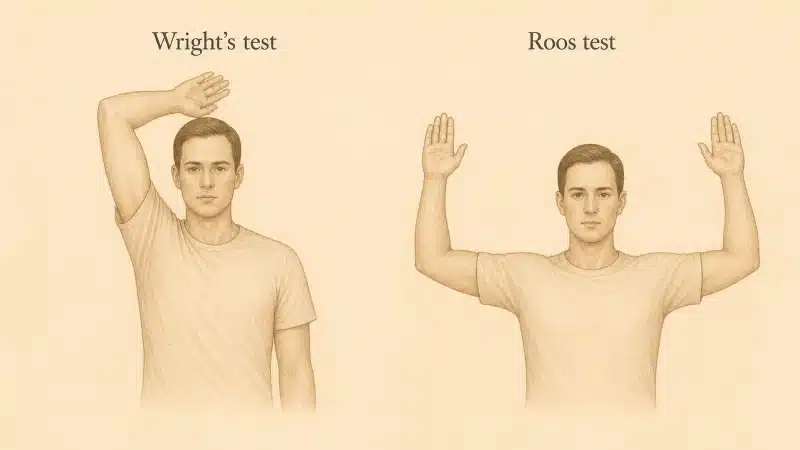
You may already have had the standard TOS workup: the Roos test (arms held overhead, opening and closing your hands for three minutes), Wright’s test (arm rotated outward and back), and imaging of your neck. If those tests reproduce the symptoms but the imaging shows no cervical rib, no obvious bony anomaly, and a normal cervical MRI, the upstream cause of TOS is unaccounted for.
In that situation, the following signs raise the question of CN XI involvement:
- Asymmetric trapezius bulk. One side of the upper shoulder looks visibly smaller or flatter than the other.
- Shoulder droop. One shoulder sits lower than the other. Often easier to see in old photographs than in the mirror.
- Lateral scapular winging. Your shoulder blade lifts off the ribcage when you raise your arm.
- Reduced shoulder shrug strength. When you shrug both shoulders against resistance, one side gives way first.
- Mild head-rotation weakness turning toward the opposite side from the affected shoulder. (This is the SCM component of CN XI weakness — most patients don’t notice it until they’re tested.)
If any of these are present alongside TOS symptoms, the question changes from “how do we treat the thoracic outlet?” to “why is the trapezius weak — and is the cause at the skull base?”
If the answer is atlanto-styloid CN XI compression, then treating only the thoracic outlet will leave the upstream problem in place. The shoulder will continue to droop. The brachial plexus will continue to be stretched. The TOS will recur.
This is one of the under-recognized causes of recurrent and persistent Thoracic Outlet Syndrome.
How CN XI Compression Is Diagnosed
Standard cervical imaging will not catch this. A proper workup needs to look at the entire chain — skull base, shoulder girdle, and thoracic outlet — rather than only one segment of it.
- CT of the skull base and upper cervical spine. Measures styloid length, the angle at which the styloid points toward C1, and the atlanto-styloid distance on both sides. This is the single most important study. A cervical-spine-only MRI will miss the problem entirely.
- CT venography. A specialized CT scan that visualizes the jugular vein, used when concurrent jugular vein compression is suspected — often the case, since both conditions share the same anatomical cause.
- MRI of the brachial plexus. To rule out other causes of TOS such as cervical rib, fibrous band, mass, or scalene anomaly.
- EMG of the trapezius and SCM. Electromyography is a test in which a thin needle records the electrical activity of muscles to determine whether the nerve supplying them is working. EMG confirms CN XI involvement and grades the severity.
- Brachial plexus nerve conduction studies. Specific electrical tests of the sensory nerves running into the arm — in particular the ulnar SNAP (sensory nerve action potential from the pinky-side sensory nerve) and the medial antebrachial cutaneous SNAP (from the inner-forearm sensory nerve). Both are characteristically reduced when the lower trunk of the brachial plexus is under chronic traction, as in neurogenic TOS, and the pattern of reduction is what distinguishes this cause from others.
Treatment: Sequence Matters
Treatment must address the entire cascade, in the right order.
1. Decompression of the atlanto-styloid corridor — first. This means transcervical styloidectomy — surgical removal of the styloid process — partial resection of the C1 transverse process, or both. The choice depends on which structure is doing most of the compressing. This is the upstream intervention. Without it, nothing downstream will stay fixed.
2. Shoulder girdle rehabilitation — second. Once the nerve is decompressed and starts to recover, the trapezius can be rebuilt with postural correction and targeted strengthening. In many patients, the shoulder droop reverses as the muscle re-innervates, and this alone resolves the brachial plexus traction.
3. Thoracic outlet decompression — only if needed. When the brachial plexus damage has become fixed — when the nerve symptoms in the arm don’t reverse with shoulder rehab — separate thoracic outlet decompression may still be required. Ideally this is staged after the upstream cause has been treated. The PURED procedure offers a minimally invasive option for patients who reach this stage.
Sequencing is the part most often gotten wrong. Operating on the thoracic outlet while leaving the styloid–C1 compression in place is one of the under-appreciated causes of recurrent TOS. The shoulder keeps drooping. The plexus keeps getting stretched. The patient comes back with the same arm symptoms a year later, and the surgical team wonders why.
A staged, integrated approach — skull base first, shoulder second, thoracic outlet last only if necessary — is usually the right answer.
Conclusion
The standard description of Eagle Syndrome captures only one face of a much broader anatomical problem. The same compression at the base of the skull that classically affects the throat nerves can also compress the spinal accessory nerve, which wraps around the internal jugular vein in the same narrow corridor.
Once CN XI fails, the trapezius weakens. The shoulder blade wings outward. The shoulder drops. The brachial plexus is placed under sustained traction at the thoracic outlet. A single bony abnormality at the atlas produces hand symptoms a meter away.
For any patient with TOS-like symptoms, a droopy shoulder, and visible trapezius weakness on one side, the right question to bring to your doctor is simple: has my skull base been imaged? A CT scan of the styloid and C1 should be done before any thoracic outlet surgery is considered.
Next Article:
PURED Procedure for Thoracic Outlet Syndrome
A minimally invasive option for the patients in whom thoracic outlet decompression is still needed after the upstream cause has been treated.
See Also:
- Eagle Syndrome: A Detailed Review
- Internal Jugular Vein Compression: A Detailed Review
- What is Thoracic Outlet Syndrome?
- Persistent and Recurrent Thoracic Outlet Syndrome
References
- Mitchell EC, Wu K, Yoo J, Ross DC, Miller TA, Siddiqi F (2023). “Winged” Eagle’s syndrome: neurophysiological findings in a rare cause of spinal accessory nerve palsy. Illustrative cases. J Neurosurg Case Lessons, 6(24):CASE23358. PMID: 38079628.
- Roberts SO, Cardozo A (2024). A detailed review of the spinal accessory nerve and its anatomical variations with cadaveric illustration. Anat Sci Int, 99(3):239–253. doi:10.1007/s12565-024-00770-w. PMID: 38696101.
- Swift TR, Nichols FT (1984). The droopy shoulder syndrome. Neurology, 34(2):212–215. PMID: 6538012.
- Lokman B, Aymane A, Yachaoui S, El Oumri AA (2024). Droopy shoulder syndrome: a gateway to thoracic outlet syndrome. Cureus, 16(6):e62213. doi:10.7759/cureus.62213. PMID: 39006624.
- Al-Shekhlee A, Katirji B (2003). Spinal accessory neuropathy, droopy shoulder, and thoracic outlet syndrome. Muscle Nerve, 28(3):383–385. PMID: 12929202.
- Mitchell EC, Wu K, Yoo J, Ross DC, Miller TA, Siddiqi F (2023). “Winged” Eagle’s syndrome: neurophysiological findings in a rare cause of spinal accessory nerve palsy. Illustrative cases. J Neurosurg Case Lessons, 6(24):CASE23358. PMID: 38079628[↩][↩]
- Roberts SO, Cardozo A (2024). A detailed review of the spinal accessory nerve and its anatomical variations with cadaveric illustration. Anat Sci Int, 99(3):239–253. doi:10.1007/s12565-024-00770-w. PMID: 38696101[↩]
- Swift TR, Nichols FT (1984). The droopy shoulder syndrome. Neurology, 34(2):212–215. PMID: 6538012[↩]
- Lokman B, Aymane A, Yachaoui S, El Oumri AA (2024). Droopy shoulder syndrome: a gateway to thoracic outlet syndrome. Cureus, 16(6):e62213. doi:10.7759/cureus.62213. PMID: 39006624[↩]
- Al-Shekhlee A, Katirji B (2003). Spinal accessory neuropathy, droopy shoulder, and thoracic outlet syndrome. Muscle Nerve, 28(3):383–385. PMID: 12929202[↩]
