If you have been told that brain fog, memory problems, or trouble concentrating are “early aging” or “early dementia,” the cause may not be inside the brain at all. It may be in the neck.
For decades, dementia research has focused almost exclusively on what happens inside the brain: the build-up of amyloid plaques, tau tangles, vascular disease of small arteries, and the gradual loss of neurons. But a growing body of evidence is now pointing to something that happens outside the skull: a mechanical problem in the neck that may starve the brain of one of its most basic needs — the ability to drain its used blood and clear its metabolic waste.
This article explains the link between Jugular Vein Compression — a mechanical narrowing of the brain’s main draining vein — and cognitive decline, including mild cognitive impairment and Alzheimer’s disease. In 2026, the first prospective surgical study addressed this directly, and the results challenge the way we think about treatable causes of dementia.
Why the Neck Matters for the Brain
To understand why a vein in the neck might affect memory, we need to step back and think about the brain as a metabolic organ.
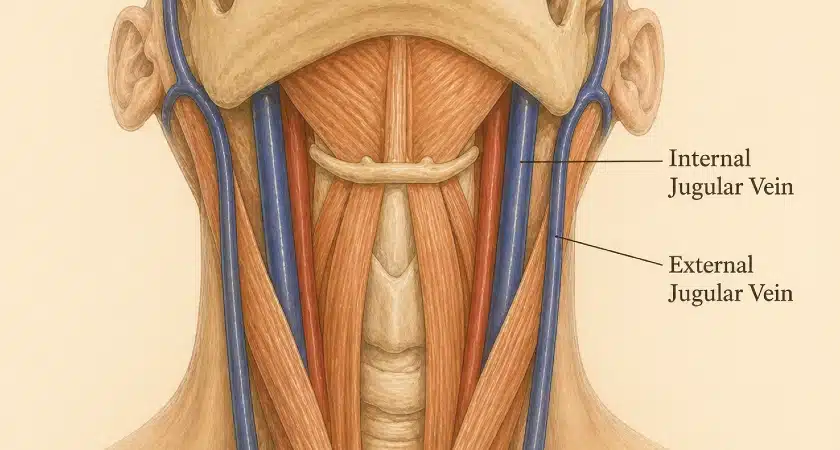
Every minute, roughly 750 ml of blood passes through the brain, delivering oxygen and removing waste products. The internal jugular veins are the main highway carrying that used blood — and dissolved metabolic waste — back toward the heart.
In stylogenic and atlanto-stylogenic Jugular Vein Compression, the jugular vein is mechanically pinched between the styloid process and the transverse process of C1 (atlas). When this anatomical “claw” closes too tightly, the vein narrows. Blood can still leave the brain — partly through the same vein, partly through smaller backup channels called the extrajugular network — but the pressure inside the skull rises and the system loses efficiency.
The recognized symptoms of jugular vein compression are headache, pulsatile tinnitus, dizziness, blurred vision, and brain fog. Recent evidence shows that the same mechanical compression also produces something more serious: measurable, progressive cognitive impairment that is often mistaken for early aging or early dementia.
What the Evidence Says
1. Patients with Alzheimer’s Disease Have Smaller Jugular Veins
In a 2025 study published in Alzheimer’s & Dementia, Pardo and colleagues compared the cross-sectional areas of the internal jugular and non-jugular drainage veins in patients with Alzheimer’s disease, mild cognitive impairment, and cognitively healthy controls. 1 The result was striking: both jugular and non-jugular veins were significantly narrower in cognitively impaired patients.
The authors proposed that a smaller venous drainage system may impair the clearance of amyloid-β42 (Aβ42) — the toxic protein at the heart of Alzheimer’s pathology — and that venous and glymphatic dysfunction together may form a vicious cycle that accelerates neurodegeneration. 1
2. Jugular Venous Reflux Predicts Cognitive Decline and Future Dementia
In a prospective Japanese study by Adachi and colleagues from Tokyo Women’s Medical University, 302 patients underwent duplex ultrasound of the jugular veins and were followed for a median of 5.2 years. 2 Patients with spontaneous jugular vein reflux scored significantly lower on both the Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA), with particular weakness in executive function and memory. Of those who developed incident dementia during follow-up, the vast majority were diagnosed with Alzheimer’s disease. 2
A separate 2025 study using ultrasound to evaluate IJV outflow in 106 patients with subjective memory complaints reached a similar conclusion: patients with impaired IJV drainage had nearly a fourfold higher odds of cognitive impairment (OR 3.84) after adjusting for age, education, and other vascular risk factors. 3
3. Animal Studies Show a Causal Link
In 2019, Fulop and colleagues demonstrated that surgically induced cerebral venous congestion in mice produced blood-brain barrier disruption, neuroinflammation, and measurable cognitive impairment. 4 A 2023 study by Wei and colleagues confirmed that jugular vein ligation in animals altered brain metabolite profiles and impaired learning and memory — establishing that obstructed venous outflow is not merely a marker of disease but can directly cause cognitive dysfunction. 5
4. Case Reports of Reversal After Jugular Decompression
Perhaps the most provocative early signal came from a 2022 case report in Neurology by Primiani and colleagues from Johns Hopkins. The authors described a patient with cerebral venous congestion and progressive cognitive decline whose symptoms improved after surgical jugular release. 6 This single case prompted a broader question that had been waiting in the wings: if compression causes the decline, can surgical release reverse it?
The 2026 Beijing Study: First Evidence That Decompression Can Improve Cognitive Scores
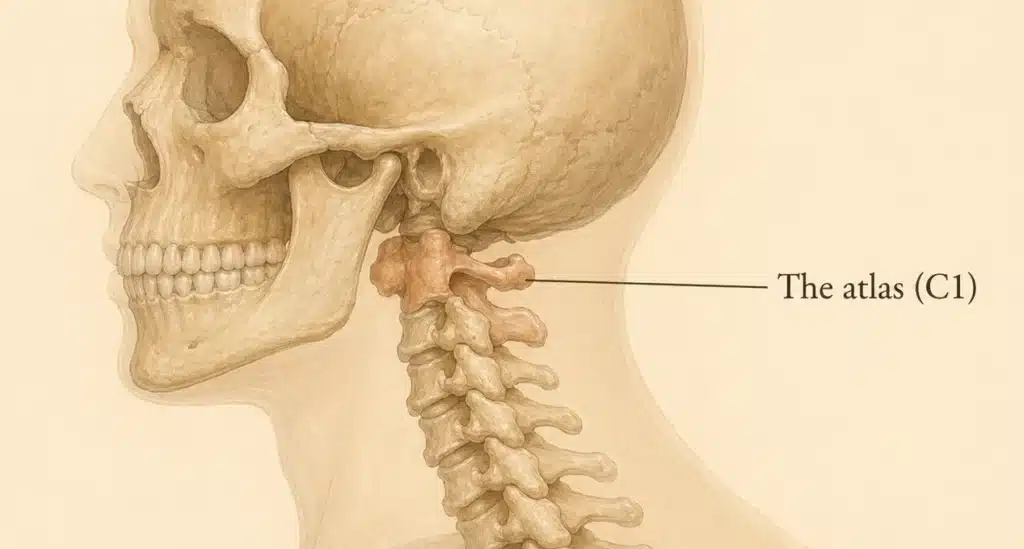
The most important development came in March 2026, when Peng and colleagues from Beijing Shijitan Hospital published the first prospective surgical study specifically targeting internal jugular vein stenosis caused by atlas (C1) transverse process compression. 7
The study enrolled 47 patients with CT venography-confirmed IJV stenosis caused by the C1 transverse process. Patients with known causes of cognitive decline (stroke, prior brain hemorrhage, Alzheimer’s diagnosis) were excluded, isolating the vein compression as the variable of interest. 7
The findings were remarkable:
- The mean MoCA score in the cohort was 23.21, well below the normal cutoff of 26 — indicating that patients with C1-induced IJV stenosis had measurable cognitive impairment even before surgery. 7
- The degree of stenosis correlated tightly with cognitive performance. A higher relative stenosis (greater percentage reduction in lumen area) predicted lower MoCA scores. The correlation remained strong (partial r = −0.66, p < 0.001) even after controlling for age and education. 7
- Sixteen patients completed a 3-month postoperative reassessment after resection of the C1 transverse process. The mean MoCA score improved from 22.56 to 24.68 (p < 0.001), with the largest gains in visuospatial/executive function and delayed recall — the same domains most affected before surgery. 7
- Symptoms such as headache, dizziness, sleep disturbance, and head noise also improved at 3 months. 7
This is, to date, the first direct clinical evidence that surgical decompression of the jugular vein at C1 can improve cognitive function. The pattern of cognitive deficits before surgery, and the matching pattern of improvement afterward, strongly suggests that the venous compression was driving the cognitive symptoms — not merely associated with them.
The authors acknowledged limitations: the sample size was small, follow-up was short, and a non-surgical control group was not included. 7 Nonetheless, the consistency of the findings with everything we already know about cerebral venous physiology makes this a result that deserves serious clinical attention.
How Jugular Vein Compression Damages the Brain
The connection between IJV compression and cognitive decline operates through at least three well-described biological pathways.
Mechanism 1: Chronic Cerebral Venous Congestion
When the jugular outflow is compressed, blood backs up inside the skull. Over time, this chronic venous congestion causes vasogenic edema, disrupts the blood-brain barrier, and provokes low-grade neuroinflammation. 4 Cerebral perfusion suffers, and the brain operates in a state of mild but constant metabolic stress. White matter tracts — particularly those running alongside the deep medullary veins — are especially vulnerable, which may explain why white matter hyperintensities on MRI correlate so strongly with cognitive decline in older adults. 8
Mechanism 2: Intracranial Hypertension and Reduced CSF Reabsorption
The cerebrospinal fluid that bathes the brain is reabsorbed into the venous system through the arachnoid granulations. When venous pressure rises because the jugular veins are obstructed, CSF reabsorption slows down, and intracranial pressure climbs to a new, higher equilibrium. This process is the same one I describe on my main [Jugular Vein Compression page] under intracranial hypertension and pseudotumor cerebri — but its long-term effect on cognition has only recently been appreciated.
Mechanism 3: Glymphatic Failure and Amyloid Accumulation
This is perhaps the most concerning mechanism. The glymphatic system is the brain’s recently discovered waste-clearance network. Cerebrospinal fluid enters the brain along the perivascular spaces around arteries, exchanges with interstitial fluid, picks up metabolic waste — including amyloid-β and tau — and exits along the perivenous spaces around the brain’s veins. 9 From there, the waste leaves the cranium along two parallel pathways: the meningeal lymphatic vessels, which drain into the deep cervical lymph nodes 10, and the arachnoid granulations, which absorb cerebrospinal fluid into the dural venous sinuses and ultimately into the internal jugular vein. 9
Both pathways are mechanically and physiologically dependent on the venous environment of the upper neck. The arachnoid-granulation route requires a low-pressure dural sinus system, which in turn requires unobstructed jugular outflow. The lymphatic route runs alongside the jugular vein and is influenced by surrounding venous pressure. When the IJV is chronically compressed at C1, several things can go wrong:
- Reduced venous outflow slows the perivenous CSF efflux that drives glymphatic clearance.
- Backup of pressure in the venous system reduces the gradient that pulls waste out of the brain.
- Amyloid-β clearance is impaired, allowing the protein to accumulate over years and decades.
This is the vascular-glymphatic hypothesis of Alzheimer’s disease: that the disease is not purely a problem of overproduction of amyloid, but of failed clearance — and that the venous system in the neck is one of the bottlenecks [1, 9].
When Cognitive Symptoms Point to Jugular Vein Compression
Not every patient with cognitive complaints has jugular vein compression — and not every patient with jugular vein compression will progress to dementia. But certain features should prompt a closer look at the venous system.
The strongest single clue is the company the cognitive symptoms keep. Brain fog, memory problems, and executive dysfunction that arrive on their own can have many causes — but when the same patient also has chronic headaches that are worse first thing in the morning or after lying down, pulsatile tinnitus (a pulsing or whooshing sound in one ear), pressure inside the head, or visual disturbances, the picture starts to look mechanical rather than neurodegenerative. These are the symptoms of cerebral venous congestion, and when they cluster together with cognitive complaints, the venous system in the neck deserves a serious look.
Demographics matter too. A patient in their late 40s or 50s with progressive memory complaints, no family history of Alzheimer’s disease, and no typical vascular risk factors is a different clinical picture than an 80-year-old with a strong family history. The first picture is much more likely to have a treatable cause underneath it.
A previous diagnostic label is also worth taking seriously. A patient who has been told they have idiopathic intracranial hypertension — high pressure inside the skull “with no known cause” — and who is now also experiencing memory or executive function problems is one of the clearest signals that the cognitive complaints and the pressure problem share the same mechanical cause. The “idiopathic” label rarely turns out to be idiopathic on close imaging.
Two further clues are worth knowing. First, symptoms that clearly worsen when lying flat and improve when the head is elevated point toward a venous drainage problem in a way that almost nothing else does. Second, an MRI showing white matter hyperintensities out of proportion to the patient’s age and vascular risk factors is a quiet signal the brain has been working under venous strain for years. Neither finding is diagnostic on its own, but either one in combination with the symptom cluster above raises the index of suspicion meaningfully.
If several of these apply, the next step is dedicated imaging of the venous system — specifically a CT venography focused on the styloid–C1 region. The full diagnostic workup is described on my Jugular Vein Compression page.
Why This Has Been Missed for So Long
If the link between jugular vein compression at C1 and cognitive decline is plausible — and the evidence is now growing — why has this not been part of mainstream dementia evaluation? There are several reasons, and they mirror the broader story of jugular vein compression as a clinical entity.
Standard Dementia Work-Ups Don’t Image the Jugular Veins
Brain MRI looks at the brain. Carotid Doppler looks at the arteries. The relationship between the styloid process, the C1 transverse process, and the jugular vein at the level just below the skull base is rarely examined in a routine dementia work-up. Without CT venography (CTV) focused on the styloid–C1 region, the compression is invisible.
Most Specialists Are Unfamiliar With the Condition
Jugular vein compression at C1 only became a recognized entity in the early 2010s. Even now, awareness is largely limited to a small group of vascular neurosurgeons and interventional neuroradiologists. Most general neurologists evaluating cognitive complaints have never been trained to consider it. 11
The Symptoms Are Non-Specific
Brain fog, slowed thinking, poor memory, and difficulty concentrating are routinely attributed to age, stress, depression, sleep apnea, or “early Alzheimer’s” without further investigation. When patients report that their symptoms worsen when lying down or improve with head elevation (a strong clue toward a venous mechanism), the clue is often dismissed.
Routine Imaging Often Appears Normal
A standard brain MRI in a patient with C1-induced IJV Compression can be entirely normal, or show only mild non-specific white matter changes, while a properly performed CT venography would clearly demonstrate the compressed vein and the C1–styloid relationship behind it.
The result of all of this is that patients with potentially treatable cognitive dysfunction are sometimes given a permanent diagnostic label and sent home to manage decline, when the actual problem is mechanical and reversible.
Can Surgery Help?
The evidence that jugular decompression at C1 can improve cognition is encouraging but still early. The 2026 Beijing study is small, the follow-up was 3 months, and there was no surgical control group. 7 Whether the cognitive gains persist long-term, whether they translate into a reduced risk of progression to Alzheimer’s disease, or which patients benefit most are all open questions.
What the current evidence supports:
- In symptomatic patients with clear C1-related jugular vein compression, surgical decompression — performed by a surgeon with specific experience in this anatomy — has been reported to improve a wide spectrum of symptoms, including headache, pulsatile tinnitus, brain fog, and now measurable cognitive performance.
- Surgery is not a cure for established Alzheimer’s disease. No reasonable clinician should claim otherwise. But in selected patients whose cognitive symptoms are driven primarily by venous congestion, the outlook is meaningfully different from neurodegenerative dementia: the cause is mechanical, and the mechanical problem can be addressed.
- The earlier the recognition, the better. Decades of unrelieved venous congestion likely produce cumulative damage in the form of white matter injury and amyloid accumulation that is harder to reverse.
For details on how surgical decompression is performed, see my page on Jugular Vein Compression Treatment.
A Word of Caution and a Word of Hope
A word of caution. Not every patient with memory complaints has a treatable mechanical problem. Most cases of dementia are not caused by jugular vein compression, and surgery is never the right answer for someone whose imaging does not clearly demonstrate the condition. Diagnosis must be objective — based on dedicated CT venography and a careful clinical correlation — not based on hope alone.
A word of hope. For a subset of patients — and I am increasingly convinced this subset is larger than once believed — chronic cognitive symptoms are not the beginning of inevitable decline. They are the consequence of a brain that is not draining properly, working harder than it should, and slowly losing its ability to clear its own waste. In these patients, the diagnosis of “early dementia” or “idiopathic intracranial hypertension” or “chronic migraine with cognitive features” can hide a problem that is mechanical, identifiable, and surgically correctable.
The relationship between the styloid process, the C1 vertebra, and the jugular vein is, at the end of the day, an anatomical relationship. And anatomical problems have anatomical solutions.
The brain is not an isolated organ. It depends on a clean, low-resistance pathway out through the neck, and when that pathway is obstructed at the level of C1, the consequences may extend far beyond a headache.
Next Article
Jugular Vein Compression Treatment: How Surgical Decompression is Performed
See Also
- Jugular Vein Compression: A Detailed Review
- Jugular Vein Compression by the Carotid Artery
- Eagle Syndrome: A Detailed Review
- Eagle Syndrome and Jugular Vein Compression: An Expert Interview
References
- Pardo K, Khasminsky V, Keret O, Benninger F, Goldberg I, Shelef I, et al. (2025). Alzheimer’s disease patients have smaller venous drainage system compared to cognitively healthy controls. Alzheimer’s & Dementia, 21:e14551. doi:10.1002/alz.14551[↩][↩]
- Adachi U, Toi S, Hosoya M, Hoshino T, Seki M, Yoshizawa H, Tsutsumi Y, Maruyama K, Kitagawa K (2023). Association of age-related spontaneous internal jugular vein reflux with cognitive impairment and incident dementia. Journal of Alzheimer’s Disease. doi:10.3233/JAD-230771[↩][↩]
- Wang YH, et al. (2025). Ultrasound evaluation of internal jugular venous insufficiency and its association with cognitive decline. Diagnostics, 15(11):1427[↩]
- Fulop GA, Ahire C, Csipo T, Tarantini S, Kiss T, Balasubramanian P, et al. (2019). Cerebral venous congestion promotes blood-brain barrier disruption and neuroinflammation, impairing cognitive function in mice. GeroScience, 41:575–589. doi:10.1007/s11357-019-00110-1[↩][↩]
- Wei H, Jiang H, Zhou Y, Xiao X, Zhou C, Ji X (2023). Cerebral venous congestion alters brain metabolite profiles, impairing cognitive function. Journal of Cerebral Blood Flow & Metabolism, 43:1857–1872. doi:10.1177/0271678X231182244[↩]
- Primiani CT, Lawton M, Hillis AE, Hui FK (2022). Pearls & Oy-sters: Cerebral venous congestion associated with cognitive decline treated by jugular release. Neurology, 99:577–580. doi:10.1212/WNL.0000000000201037[↩]
- Peng X, Xu J, Lu S, Ma H, Xu S, Lv M, Hu Z, Ding Y, Ji X, Zhu G (2026). Surgical treatment for cognitive impairment caused by internal jugular vein stenosis: a clinical study of atlas transverse process resection. Frontiers in Neurology, 17:1776658. doi:10.3389/fneur.2026.1776658[↩][↩][↩][↩][↩][↩][↩][↩]
- Kapadia A, et al. Venous dysfunction plays a critical role in “normal” white matter disease of aging. Reviewed in: Csiszar A, et al. Journal of Cerebral Blood Flow and Metabolism. 2020[↩]
- Iliff JJ, Wang M, Liao Y, et al. (2012). A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Science Translational Medicine, 4:147ra111[↩][↩]
- Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, et al. (2015). Structural and functional features of central nervous system lymphatic vessels. Nature, 523:337–341. doi:10.1038/nature14432[↩]
- Scerrati A, Norri N, Mongardi L, et al. (2021). Styloidogenic-cervical spondylotic internal jugular venous compression, a vascular disease related to several clinical neurological manifestations: diagnosis and treatment—a comprehensive literature review. Annals of Translational Medicine, 9(8):718. doi:10.21037/atm-20-7698[↩]
