For decades, idiopathic intracranial hypertension (IIH) – also known as pseudotumor cerebri – has carried a name that all but tells the patient we do not know why this is happening. The condition is defined by elevated cerebrospinal fluid (CSF) pressure in the absence of a brain tumor, hydrocephalus, or another obvious structural cause 1 Patients are typically obese, female, and of reproductive age 2 3 and they present with debilitating headaches, pulsatile tinnitus, papilledema, visual obscurations, and progressive vision loss 4
The classical narrative blames obesity, hormones, and a vague problem with CSF reabsorption. But in real clinical practice, this narrative leaves many patients without answers and without effective long-term treatment. A growing body of evidence shows that, in a substantial proportion of patients, the elevated pressure is not idiopathic at all – it is the downstream consequence of a mechanical obstruction to cerebral venous outflow 5 6 7
Two of the most under-recognized obstructions occur at very specific anatomical bottlenecks: the internal jugular vein at the level of the first cervical vertebra (C1) and the subclavian – brachiocephalic venous corridor at the thoracic outlet. Both should be on the differential of every patient labeled with IIH, and especially in those whose symptoms persist despite weight loss, acetazolamide, lumbar punctures, CSF shunts, or even venous sinus stenting.
This article explains the rationale, the anatomy, the supporting evidence, and the practical implications.
What IIH Actually Is – A Brief Refresher
The current diagnostic standard is the 2013 Friedman criteria, which require 1
- Symptoms and signs of generalized intracranial hypertension or papilledema
- Normal neurological examination apart from cranial nerve abnormalities
- Normal neuroimaging (no mass, hydrocephalus, or structural lesion)
- Normal CSF composition
- Elevated lumbar puncture opening pressure (>25 cm H₂O in adults, >28 cm H₂O in children)
If any cause – dural sinus thrombosis, certain medications, endocrine disease – is identified, the case is reclassified as secondary intracranial hypertension and the “idiopathic” label is dropped. The problem is that under the current paradigm, clinicians rarely look beyond the skull for venous obstruction. Standard imaging stops at the skull base, the patient is supine and head-neutral during scanning, and the dynamic narrowings that only manifest in the upright posture or with head rotation are invisible.
The result is predictable: a patient with a perfectly identifiable mechanical cause is labeled idiopathic and treated symptomatically.
Classical demographics still apply – but they don’t tell the whole story
- IIH affects women approximately 9 times more often than men 3
- Peak incidence is between 20 and 40 years of age 3
- The strongest classically recognized risk factor is obesity, present in 70–94% of patients 2
- Up to 40% develop persistent visual impairment, and 1–10% become legally blind 3
These numbers are real, and obesity does play a role – elevated intra-abdominal and central venous pressures genuinely increase intracranial venous pressure 5 But obesity alone does not explain the lean IIH patient, the patient with refractory disease despite weight loss, or the patient with idiopathic intracranial hypertension without papilloedema (IIHWOP) 8
The Jugular Hypothesis – A Unifying Model
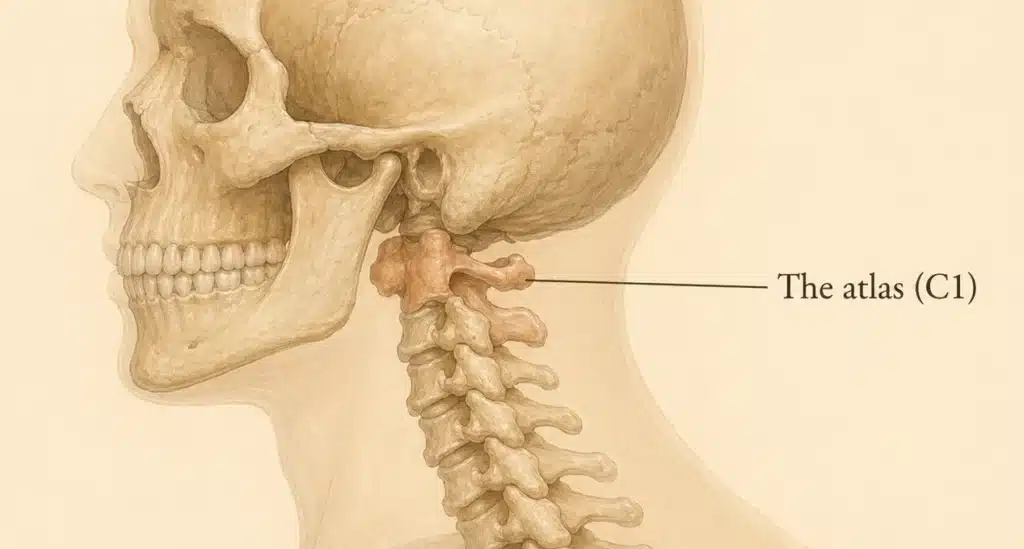
In 2024, Fargen, Midtlien, Margraf, and Hui published the most important reframing of IIH pathogenesis in recent memory: the jugular hypothesis 5 Their central argument is that IIH and its spectrum disorders are best understood as a problem of cerebral venous outflow, with the internal jugular veins (IJV) as the primary, vulnerable, and frequently overlooked culprit.
Their model can be summarized in six points 5
- The IJV are prone to physiological and pathological compression along their entire course – at C1 by the styloid process, and between C3 and C6 by dynamic muscular and carotid forces.
- Severe dynamic IJV stenosis with large cervical pressure gradients is common in IIH-spectrum patients and significantly impacts intracranial venous and CSF pressures.
- Extra-jugular collateral pathways dilate over time, but they are insufficient to substitute for normal jugular outflow.
- Poor IJV outflow drives intracranial venous hypertension and congestion, raising CSF pressures and intracranial pressure (ICP).
- The glymphatic system also becomes congested but cannot compensate.
- Persistently elevated ICP eventually produces transverse sinus extramural compression – the classic finding seen on imaging – which acts as an amplifier, not the primary cause.
This last point is critical and routinely misunderstood in clinical practice. Transverse sinus stenosis on MR venography is the finding that has dominated IIH workups for two decades, while the much more proximal – and physiologically more important – obstruction at the internal jugular vein rarely makes it into the imaging report at all. Clinical attention is fixed on the downstream consequence rather than the upstream cause. The chicken-or-egg question was first formally framed by Corbett and Digre in 2002 9 and the answer has since become reasonably clear. King and colleagues showed in the same year that lowering CSF pressure by lateral C1-C2 puncture immediately resolves the elevated venous sinus pressure measured during direct manometry 10– an observation that is fundamentally incompatible with the stenosis being the upstream culprit. Subsequent imaging studies have repeatedly confirmed the same finding: transverse sinus stenoses reverse on catheter and CT venography, sometimes within an hour, after CSF withdrawal or shunting 11
To be objective, the reversibility is not universal. Bono and colleagues followed fourteen IIH patients with bilateral transverse sinus stenosis on serial MR venography over six years and found that the stenoses persisted even in the nine patients (64%) whose CSF pressure normalized on medical treatment 12– suggesting that in a subset of cases the sinus undergoes structural remodeling that no longer responds to ICP reduction alone. De Simone and colleagues synthesized these mixed observations into a self-limiting venous collapse feedback-loop model, in which an initial rise in CSF pressure compresses a collapsible sinus segment, the resulting venous pressure rise further reduces CSF reabsorption, and both pressures stabilize at a new higher equilibrium 13 The model accommodates both patterns: in some patients the loop is interrupted by acute CSF withdrawal and the stenosis reverses; in others, particularly with prolonged disease, the equilibrium becomes entrenched and the stenosis persists despite pressure normalization. Either way, the transverse sinus stenosis is a self-amplifying participant, not the original cause.
This is precisely why a substantial number of patients relapse after venous sinus stenting. A 2024 systematic review and meta-analysis of 694 IIH patients across 24 studies reported a pooled restenosis rate of 17.7% and found that 22.3% of patients had persistent or worsened symptoms after stenting; the authors concluded that these rates highlight the need for further investigation of the procedure 14 An earlier cohort study by Ahmed and colleagues followed 52 IIH patients after transverse sinus stenting and reported that 6 patients (11.5%) had symptom relapse driven by both elevated venous pressure and a new stenosis immediately adjacent to the original stent, requiring a second stenting procedure; one patient ultimately needed four 15 Stenting splints open the consequence while the upstream obstruction – at the jugular vein or further downstream – is left entirely untouched 5
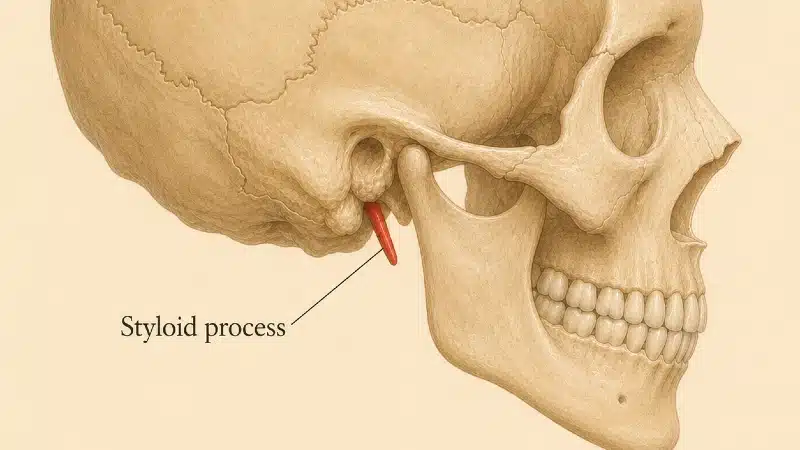
Compression Point #1: Internal Jugular Vein Stenosis at C1 (Styloidogenic Jugular Venous Compression)
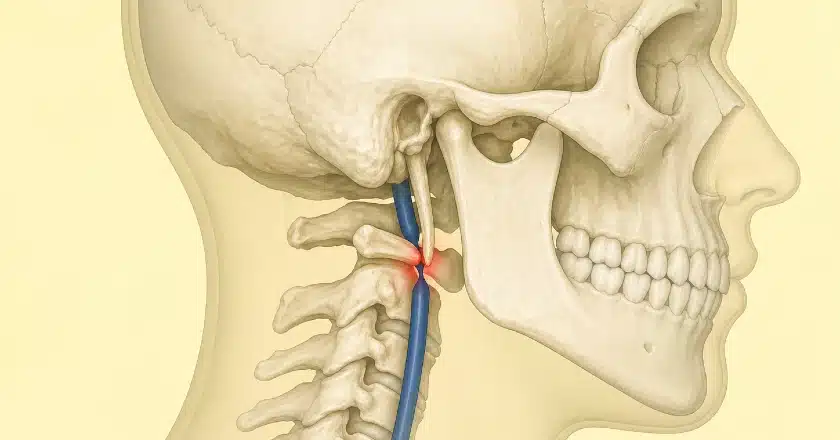
The internal jugular vein leaves the skull through the jugular foramen and immediately enters a narrow corridor between two hard structures: the styloid process of the temporal bone in front, and the transverse process of the C1 (atlas) vertebra behind. In some individuals, this corridor is so narrow that the vein is mechanically pinched – a phenomenon known as styloidogenic jugular venous compression syndrome 16 17 18 19
Think of it as a garden hose squeezed between two rocks. Even a relatively normal-length styloid can produce dramatic narrowing if it is angled inward or if the C1 transverse process is unusually large or laterally placed 18 Soft-tissue contributors – the digastric muscle, the occipital artery looping around the vein, the rectus capitis muscles, fibrous bands within the carotid sheath – further compound the obstruction 19
I have discussed the full anatomy, symptomatology, and surgical management of this entity on a dedicated page on this site: Jugular Vein Compression.
Why C1 compression matters for IIH
When the IJV is compressed at C1, blood backs up into the dural venous sinuses. The brain’s venous pressure rises. Because CSF reabsorption depends on a roughly 3 to 5 mmHg pressure gradient between CSF and venous sinus blood 5 any rise in venous pressure forces a compensatory rise in CSF pressure. The patient develops headaches, pulsatile tinnitus, papilledema, visual obscurations, brain fog – and is labeled IIH.
Multiple groups have now demonstrated that relieving the jugular obstruction resolves the intracranial hypertension in carefully selected patients, with normalization of opening pressures and resolution of symptoms 20 Surgical decompression – removal of the styloid process, the C1 transverse process, or both, with release of any contributing soft-tissue compressors – is the most logical and durable approach, because it addresses the actual mechanical cause rather than splinting the vein open against an unrelenting external force.
The clue most clinicians miss: positional symptoms
C1 jugular compression is dynamic. It worsens with neck motion, and – because the IJV is the dominant cerebral drainage route in recumbency – when the patient lies flat. A patient whose headache is worst on waking, who sleeps semi-upright by instinct, whose tinnitus disappears with manual neck pressure, who notices brain fog after head rotation, is describing mechanical features rather than migraine features.
Compression Point #2: The Thoracic Outlet – A Forgotten Layer
This is where the conversation becomes more interesting, and where the literature has only recently begun to catch up.
The internal jugular vein does not drain directly into the heart. It joins the subclavian vein behind the medial end of the clavicle to form the brachiocephalic (innominate) vein, which then crosses the upper mediastinum to enter the superior vena cava. This entire stretch – from the medial clavicle to the SVC – runs through the thoracic outlet, the same anatomical bottleneck that produces Thoracic Outlet Syndrome (TOS).
Most clinicians think of TOS as a brachial plexus or subclavian artery problem. That is the standard teaching, and it is correct as far as it goes – neurogenic TOS accounts for 80–85% of cases. But the venous component is more pervasive than the textbooks suggest, and its consequences extend far beyond the arm.
The extrajugular venous network
The internal jugular veins are the dominant cerebral venous drainage route in supine posture. In the upright posture, however, the IJVs sit above heart level and collapse under atmospheric pressure, ceasing to function as the primary outflow channel 21 When this happens, cerebral venous blood is shunted into an alternative system: the extrajugular venous network (EJN).
The EJN comprises the vertebral venous plexus (internal and external), the deep cervical veins, the external jugular vein, and the emissary veins of the posterior cranial fossa. At the skull base, the two pathways are anatomically linked through the Anterior Condylar Confluence (ACC) of Trolard – a venous hub at the external orifice of the hypoglossal canal that connects the jugular bulb directly to the vertebral plexus via the condylar veins 22 Mathematical modeling and upright imaging studies have demonstrated that in the upright position the vertebral plexus becomes the predominant outflow pathway for cerebral venous blood, while the IJVs carry a minor share 21 23 The two systems function as parallel circuits that converge at the same anatomical termination: the subclavian and brachiocephalic veins at the base of the neck.
How TOS overwhelms cerebral venous outflow
The convergence of these two drainage systems at the neck base is what links thoracic outlet syndrome to intracranial pressure. When the subclavian vein is chronically compressed at the costoclavicular junction, blood from the upper extremity cannot return through the obstructed segment and is forced into collateral pathways.
The systematic imaging literature documents precisely where these collaterals go. In Marini and colleagues’ pictorial review of thoracic collateral patterns, the cervical venous channels are identified as the primary collateral territory for axillary and subclavian obstruction 24 The named anterior neck pathways include the internal jugular vein, external jugular vein, jugular venous arch, inferior thyroid vein, and vertebral venous plexus. Posterior neck collaterals – described as more common and more pronounced – run through the same vertebral plexus and may either cross to the contralateral side or drain into the chest wall and azygos system 24
These collateral channels are precisely the vessels that comprise the extrajugular venous network draining the brain. In a patient with venous TOS, the EJN is therefore performing two functions simultaneously: draining the upper extremity through retrograde collateral flow, and draining the brain. In recumbency, the IJVs are open and carry the majority of cerebral venous return, and this dual demand may be tolerated. In the upright posture, when the IJVs collapse and the brain becomes physiologically dependent on the extrajugular network, the system is overwhelmed. Cerebral venous blood cannot drain efficiently through channels already congested with retrograde subclavian collateral flow. The consequence is impaired cerebral venous outflow, elevated cerebral venous pressure, and – through the pressure-dependent mechanism of CSF reabsorption – elevated intracranial pressure.
This redirection of flow has been captured directly on imaging. In 2017, Chahwala and colleagues described a patient with debilitating migraines preceded by unilateral arm swelling. On dynamic venography, with arm adduction, contrast injected into the subclavian vein flowed retrograde into the ipsilateral internal jugular vein, with extensive collateral channels diverting blood toward the head 25 Her migraines resolved completely after scalenectomy and first rib resection. This case is the most direct angiographic demonstration in the published literature that costoclavicular venous obstruction can force arm-derived venous return into the cerebral outflow pathway.
The high prevalence of IJV abnormalities in TOS patients on routine venography is consistent with this physiology. In a prospective venographic study of consecutive patients with neurogenic TOS, Ahn and colleagues found internal jugular vein stenosis in 76.2% (47.6% bilateral) alongside subclavian vein stenosis in 82.5% 26 An IJV recruited as a retrograde collateral for an obstructed subclavian system would be expected to appear narrowed and surrounded by abnormal collaterals on contrast injection, whether the underlying problem is fixed compression or functional congestion.
Forty years of clinical evidence – TOS surgery improves intractable headaches
The argument that TOS contributes to elevated intracranial pressure is not new. It is, in fact, four decades old – but the literature has remained scattered across vascular surgery, neurology, and neuro-ophthalmology, and has not yet been synthesized into the framework it deserves.
In 1985, Raskin, Howard, and Ehrenfeld followed thirty consecutive TOS patients for at least six months after transaxillary first rib resection. Twenty-six of the thirty had reported recurring headache preceding the appearance of neck and shoulder pain. After surgery, thirteen patients became completely headache-free, eleven previously disabled patients returned to work, and a further eleven reported new responsiveness to vasoactive medications that had been ineffective preoperatively 27 At the time, the mechanism was attributed to vague “vascular factors.” We now have a clearer angiographic picture of what those factors look like, as Chahwala’s 2017 case described above demonstrated.
The most recent and largest series, published by Cha and colleagues in 2025 in Clinical Anatomy, examined fifty consecutive patients carrying the dual diagnosis of chronic migraine and TOS 28 The clinical features were illuminating:
- Headaches were side-locked or more severe ipsilateral to the TOS limb in 76% of patients
- Limb swelling co-existed in 67%
- And – the most diagnostically useful clue – 84% reported headaches that were worsened by recumbency, exactly the pattern expected when intracranial drainage becomes increasingly dependent on the jugular pathway during sleep
Thirty-two of the fifty patients underwent surgical decompression, with substantial improvement in headache burden 28 The recumbent worsening is the bedside fingerprint of cerebral venous outflow disease. Patients describe waking with headaches, sleeping semi-upright by instinct, and finding that head elevation provides more relief than medication does.
The pieces fit together into a single coherent story spanning forty years. Raskin’s 1985 clinical observation of headaches resolving after first rib resection 27 Ahn’s 2014 anatomical proof that 76% of nTOS patients carry IJV stenosis 26 Chahwala’s 2017 angiographic demonstration of retrograde flow into the cerebral venous system from a TOS-level obstruction 25 Cha’s 2025 modern case series documenting the bedside features and surgical responsiveness in fifty patients 28 Each was published in a different specialty journal, and the field has not yet synthesized them. That synthesis is what this article attempts.
Putting It Together: A Two-Level Cerebral Venous Outflow Model
When IIH is reframed as a venous outflow disease, the clinical picture clarifies considerably. Cerebral venous blood must travel a gauntlet:
- From the dural sinuses, through the jugular foramen
- Past the styloid – C1 corridor (compression point 1)
- Down through the dynamic muscular and carotid relationships of C3 – C6
- Across the costoclavicular space at the thoracic outlet (compression point 2)
- Through the brachiocephalic vein and the SVC into the right atrium
A bottleneck at any level raises pressure upstream, and bottlenecks at multiple levels compound the problem. Because the system is dynamic – affected by head position, arm position, respiration, and posture – static supine imaging routinely misses these stacked obstructions.
This explains some of the most frustrating observations in IIH practice:
- Why CSF-diversion procedures and pressure-lowering medications often provide only temporary relief – they don’t address the upstream mechanical obstruction
- Why some patients relapse after weight loss (mechanical obstruction is unaffected by BMI)
- Why “idiopathic” intracranial hypertension overlaps so heavily with TOS, Eagle syndrome, POTS, EDS, and CSF leak phenotypes 29 30
- Why papilledema can be absent (IIHWOP) when collaterals partially compensate 8
Diagnostic Implications – What Should Be Imaged
If a patient presents with the IIH phenotype, the workup should not stop at brain MRI and a lumbar puncture. A complete venous outflow evaluation should include:
- CT venography (CTV) of the head and neck – the current gold standard for visualizing the styloid – C1 corridor and quantifying jugular stenosis 16 17
- MR venography (MRV) – useful for flow patterns and collateral assessment 5
- Dynamic imaging – imaging in different head positions and, where possible, in the upright posture, since static supine images underestimate dynamic stenosis 5
- Catheter venography with pressure measurements – the only reliable way to quantify trans-stenotic gradients; gradients of >3–5 mmHg across a stenosis are clinically significant 20
- Brachiocephalic and subclavian venography – particularly if the patient has any arm symptoms, has clinical features of TOS, or has not responded to upstream interventions 26
- Provocative maneuvers – arms-up MRV/MRA can reveal costoclavicular venous compression that resting imaging misses
A thorough history is at least as important as the imaging. Positional headaches, head-rotation tinnitus, neck pressure, arm symptoms, post-exertional brain fog, recumbent worsening of headache, and a personal or family history of hypermobility all push the differential toward a mechanical venous outflow problem.
Treatment Implications
Treatment must match the cause. A few principles follow from the venous outflow model:
- Acetazolamide and weight loss lower CSF production and central venous pressure respectively. They help, but they do not fix mechanical obstruction.
- Lumboperitoneal and ventriculoperitoneal shunts address the symptom (high CSF pressure) without addressing the cause. Long-term failure rates are high.
- Surgical decompression of stylogenic IJV compression – removal of the styloid process, the C1 transverse process, or both, with release of soft-tissue compressors – directly addresses the upstream cause. Selected case series show normalization of intracranial pressure and resolution of papilledema 16 20 The treatment overview is on the Jugular Vein Compression Treatment page.
- First rib resection and thoracic outlet decompression address the downstream component when the costoclavicular space is the limiting bottleneck. Forty years of clinical data – from Raskin’s original 1985 series through Cha’s 2025 case series – document substantial headache improvement after this procedure in appropriately selected patients 27 28 The PURED procedure I have developed allows complete first rib resection from a posterior approach with substantially better access to the venous and neural structures than traditional supraclavicular or transaxillary approaches.
- Sequential, staged decompression is sometimes necessary in patients with stacked compressions. The principle is the same in vascular surgery generally: relieve the most hemodynamically significant lesion first, reassess, then address the next.
Not every IIH patient needs surgery. But every IIH patient deserves a workup that can identify a mechanical cause when one is present, and a substantial fraction have one.
Reframing the IIH workup
The label “idiopathic intracranial hypertension” should be understood for what it is: a placeholder that reflects the limits of the standard workup, not a final diagnosis. When the venous outflow tract is examined carefully – from the jugular foramen, through the C1 corridor, down the dynamic mid-cervical course, and across the thoracic outlet – a mechanical cause is identifiable in a substantial fraction of patients 5 20 26
C1 jugular vein compression and thoracic outlet syndrome are two of the most commonly missed contributors. They share a common patient phenotype, often co-exist, and produce a clinical picture indistinguishable from “classical” IIH. The mechanistic argument is no longer speculative: Ahn’s anatomical work documents IJV stenosis at the thoracic outlet in three of every four nTOS patients 26 Chahwala’s case demonstrates retrograde flow into the cerebral venous system on real-time venography 25 and forty years of surgical evidence – from Raskin to Cha – document that decompressing the thoracic outlet relieves the headaches that result 27 28 Recognizing these patients requires a willingness to look beyond the skull and to image the venous outflow tract dynamically. Treating them requires expertise that bridges neurosurgery, vascular surgery, and neurointervention – exactly the multidisciplinary approach our clinic is built around.
If you have been told you have IIH, your symptoms are not improving, and no one has imaged your jugular veins or your thoracic outlet, that conversation is worth having.
Next Article
Jugular Vein Compression: A Detailed Review
See Also
- What Is Thoracic Outlet Syndrome?
- Eagle Syndrome
- PURED Procedure for Thoracic Outlet Syndrome
- Jugular Vein Compression Treatment
References
- Friedman DI, Liu GT, Digre KB. Revised diagnostic criteria for the pseudotumor cerebri syndrome in adults and children. Neurology. 2013;81(13):1159–1165. doi:10.1212/WNL.0b013e3182a55f17[↩][↩]
- Markey KA, Mollan SP, Jensen RH, Sinclair AJ. Understanding idiopathic intracranial hypertension: mechanisms, management, and future directions. Lancet Neurol. 2016;15(1):78–91. doi:10.1016/S1474-4422(15)00298-7[↩][↩]
- Mollan SP, Davies B, Silver NC, et al. Idiopathic intracranial hypertension: consensus guidelines on management. J Neurol Neurosurg Psychiatry. 2018;89(10):1088–1100. doi:10.1136/jnnp-2017-317440[↩][↩][↩][↩]
- Wall M, Kupersmith MJ, Kieburtz KD, et al. The Idiopathic Intracranial Hypertension Treatment Trial: clinical profile at baseline. JAMA Neurol. 2014;71(6):693–701. doi:10.1001/jamaneurol.2014.133[↩]
- Fargen KM, Midtlien JP, Margraf CR, Hui FK. Idiopathic intracranial hypertension pathogenesis: The jugular hypothesis. Interv Neuroradiol. 2024 Aug 8:15910199241270660. doi:10.1177/15910199241270660[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Townsend RK, Fargen KM. Intracranial venous hypertension and venous sinus stenting in the modern management of idiopathic intracranial hypertension. Life (Basel). 2021;11(6):508. doi:10.3390/life11060508[↩]
- Bono F, Messina D, Giliberto C, et al. Bilateral transverse sinus stenosis predicts IIH without papilledema in patients with migraine. Neurology. 2006;67(3):419–423. doi:10.1212/01.wnl.0000227892.67354.85[↩]
- Mollan SP, Aguiar M, Evison F, Frew E, Sinclair AJ. The expanding burden of idiopathic intracranial hypertension. Eye (Lond). 2019;33(3):478–485. doi:10.1038/s41433-018-0238-5[↩][↩]
- Corbett JJ, Digre K. Idiopathic intracranial hypertension: an answer to “the chicken or the egg?” Neurology. 2002;58(1):5–6. doi:10.1212/wnl.58.1.5[↩]
- King JO, Mitchell PJ, Thomson KR, Tress BM. Manometry combined with cervical puncture in idiopathic intracranial hypertension. Neurology. 2002;58(1):26–30. doi:10.1212/wnl.58.1.26[↩]
- Scoffings DJ, Pickard JD, Higgins JN. Resolution of transverse sinus stenoses immediately after CSF withdrawal in idiopathic intracranial hypertension. J Neurol Neurosurg Psychiatry. 2007;78(8):911–912. doi:10.1136/jnnp.2006.111765[↩]
- Bono F, Giliberto C, Mastrandrea C, Cristiano D, Lavano A, Fera F, Quattrone A. Transverse sinus stenoses persist after normalization of the CSF pressure in IIH. Neurology. 2005;65(7):1090–1093. doi:10.1212/01.wnl.0000178889.63571.e5[↩]
- De Simone R, Ranieri A, Montella S, Bilo L, Cautiero F. The role of dural sinus stenosis in idiopathic intracranial hypertension pathogenesis: the self-limiting venous collapse feedback-loop model. Panminerva Med. 2014;56(3):201–209.[↩]
- Lim J, Monteiro A, Kuo CC, Jacoby WT, Cappuzzo JM, Becker AB, Davies JM, Snyder KV, Levy EI, Siddiqui AH. Stenting for Venous Sinus Stenosis in Patients With Idiopathic Intracranial Hypertension: An Updated Systematic Review and Meta-Analysis of the Literature. Neurosurgery. 2024;94(4):648–656. doi:10.1227/neu.0000000000002718[↩]
- Ahmed RM, Wilkinson M, Parker GD, Thurtell MJ, Macdonald J, McCluskey PJ, Allan R, Dunne V, Hanlon M, Owler BK, Halmagyi GM. Transverse Sinus Stenting for Idiopathic Intracranial Hypertension: A Review of 52 Patients and of Model Predictions. AJNR Am J Neuroradiol. 2011;32(8):1408–1414. doi:10.3174/ajnr.A2575[↩]
- Mejia-Vergara AJ, Sultan W, Kostas A, Mulholland CB, Sadun A. Styloidogenic Jugular Venous Compression Syndrome with Papilloedema: Case Report and Review of the Literature. Neuroophthalmology. 2021;46(1):54–58. doi:10.1080/01658107.2021.1887288[↩][↩][↩]
- Dashti SR, Nakaji P, Hu YC, et al. Styloidogenic jugular venous compression syndrome: diagnosis and treatment: case report. Neurosurgery. 2012;70(3):E795 – E799. doi:10.1227/NEU.0b013e3182333859[↩][↩]
- Pokeerbux MR, Delmaire C, Morell-Dubois S, Demondion X, Lambert M. Styloidogenic compression of the internal jugular vein, a new venous entrapment syndrome? Vasc Med. 2020;25(4):378–380. doi:10.1177/1358863X20902842[↩][↩]
- Scerrati A, Norri N, Mongardi L, et al. Styloidogenic-cervical spondylotic internal jugular venous compression, a vascular disease related to several clinical neurological manifestations: diagnosis and treatment – a comprehensive literature review. Ann Transl Med. 2021;9(8):718. doi:10.21037/atm-20-7698[↩][↩]
- Zhou D, Meng R, Zhang X, et al. Intracranial hypertension induced by internal jugular vein stenosis can be resolved by stenting. Eur J Neurol. 2018;25(2):365-e13. doi:10.1111/ene.13512[↩][↩][↩][↩]
- Holmlund P, Johansson E, Qvarlander S, Wåhlin A, Ambarki K, Koskinen LO, Malm J, Eklund A. Human jugular vein collapse in the upright posture: implications for postural intracranial pressure regulation. Fluids Barriers CNS. 2017;14:17. doi:10.1186/s12987-017-0065-2[↩][↩]
- San Millán Ruíz D, Gailloud P, Rüfenacht DA, Delavelle J, Henry F, Fasel JH. The craniocervical venous system in relation to cerebral venous drainage. AJNR Am J Neuroradiol. 2002;23(9):1500–1508.[↩]
- Gisolf J, van Lieshout JJ, van Heusden K, Pott F, Stok WJ, Karemaker JM. Human cerebral venous outflow pathway depends on posture and central venous pressure. J Physiol. 2004;560(Pt 1):317–327. doi:10.1113/jphysiol.2004.070409[↩]
- Marini TJ, Chughtai K, Nuffer Z, Hobbs SK, Kaproth-Joslin K. Blood finds a way: pictorial review of thoracic collateral vessels. Insights Imaging. 2019;10(1):63. doi:10.1186/s13244-019-0753-3[↩][↩]
- Chahwala V, Tashiro J, Li X, Baqai A, Rey J, Robinson HR. Venous Thoracic Outlet Syndrome as a Cause of Intractable Migraines. Ann Vasc Surg. 2017;39:285.e5–285.e8. doi:10.1016/j.avsg.2016.05.109[↩][↩][↩]
- Ahn SS, Miller TJ, Chen SW, Chen JF. Internal jugular vein stenosis is common in patients presenting with neurogenic thoracic outlet syndrome. Ann Vasc Surg. 2014;28(4):946–950. doi:10.1016/j.avsg.2013.12.009[↩][↩][↩][↩][↩]
- Raskin NH, Howard MW, Ehrenfeld WK. Headache as the leading symptom of the thoracic outlet syndrome. Headache. 1985;25(4):208–210. doi:10.1111/j.1526-4610.1985.hed2504208.x[↩][↩][↩][↩]
- Cha YH, Randall L, Weber J, Ahn S. Treatment of thoracic outlet syndrome to relieve chronic migraine. Clin Anat. 2025;38(3):314–323. doi:10.1002/ca.24242[↩][↩][↩][↩][↩]
- Midtlien JP, Curry BP, Chang E, et al. Characterizing a new clinical phenotype: the co-existence of cerebral venous outflow and connective tissue disorders. Front Neurol. 2023;14:1305972. doi:10.3389/fneur.2023.1305972[↩]
- Manupipatpong S, Primiani CT, Fargen KM, et al. Jugular venous narrowing and spontaneous spinal cerebrospinal fluid leaks: A case – control study exploring association and proposed mechanism. Interv Neuroradiol. 2024;30(6):812–818. doi:10.1177/15910199241287417[↩]
